What is Spectroscopy?
We know that Niels Bohr used the science of spectroscopy to arrive at his conclusion of the atom. But what is exactly spectroscopy? Spectroscopy is a scientific measurement technique. It measures light that is emitted, absorbed, or scattered by materials. Different types of materials have colours that are only unique to them and in this way, spectroscopy can be used to study, identify and quantify those materials.
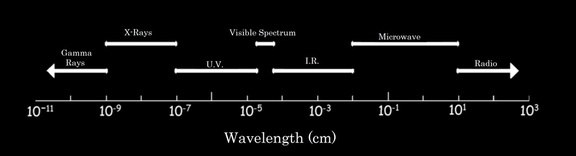
One thing that you need to remember is that "light" is a lot more than just the colored visible light that we can see. In addition to the traditional "ROYGBIV" (Red, Orange, Yellow, Green, Blue, Indigo, Violet) color spectrum, there are gamma rays, x-rays, ultraviolet (UV) radiation, infrared (IR) radiation, microwaves and radio waves.
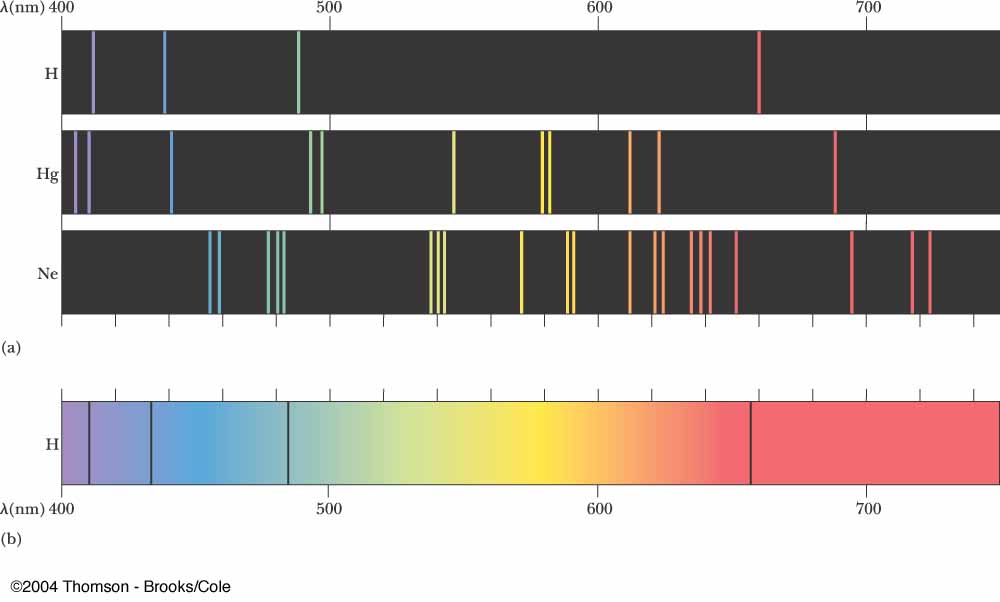
An emission spectrum in the visible light range may look like the picture below. Such a spectrum would be created when material is given extra energy somehow (it's heated, electrified, radiated with light, etc.) and that extra energy is later emitted as light energy.
What experiment did Bohr carry out?
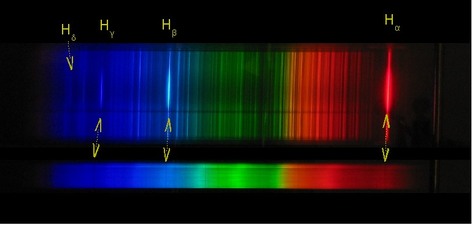
Bohr's experiment was simple. He heated hydrogen gas confined in a tube which caused the gas inside it to excite, releasing energy. When viewed through a diffraction grating, he saw that the single ray of light was actually formed by different colours. Using this information, Bohr came up with his atomic theory.