How did Rutherford discover the Nucleus?
A large portion of Rutherford's research has always included the use and study of alpha particles ever since he classified them in 1898.
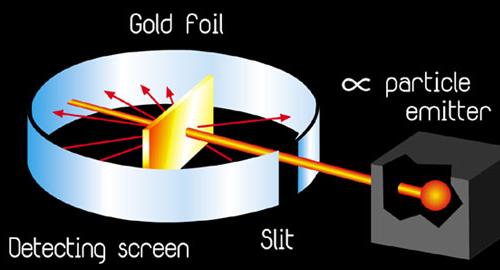
Starting sometime around 1909, Rutherford began to notice that alpha particles would not always behave in accordance to the plum pudding model of an atom when fired at a piece of gold foil. These observations stimulated further research that was eventually published in 1911 and has been known ever since as Rutherford's Gold Foil Experiment.
Throughout the course of his experiment, Rutherford had his two associates (Hans Geiger and Ernest Marsden) aim a beam of alpha particles at a piece of gold foil that was approximately 8.6 x 10^(-6) centimeters thick. To be more accurate Rutherford actually included a wide variety of different foils (such as: aluminum, iron, and lead), but his use of gold foil is most commonly spoke of. In accordance to the J.J. Thomson model of an atom, the alpha particles should have passed directly through the gold foil for all instances. Therefore to confirm this activity, a zinc sulfide screen was placed behind the foil as a backdrop for the alpha particles to appear upon. Directly above this screen was a microscope that allowed one of the two experimenters to observe any contact made between the alpha particles and the screen.
After the experiment had been set up in accordance to the speculations described above, a beam of alpha particles was fired through the piece of foil and the location where the particle landed on the screen. As explained above, each particle should have gone directly through the foil if the plum pudding model was correct. For the most part, the alpha particles corresponded with this hypothesis and passed straight through the gold foil. There were, however, a small hand full of particles that deflected slightly from the straight path by about one or two degrees. But the biggest discovery was made when 1 in 20,000 particles would deflect approximately 90 degrees or more from the parent beam. In fact, an occasional particle even fired right back at the experimenter. About this, Rutherford said:
Starting sometime around 1909, Rutherford began to notice that alpha particles would not always behave in accordance to the plum pudding model of an atom when fired at a piece of gold foil. These observations stimulated further research that was eventually published in 1911 and has been known ever since as Rutherford's Gold Foil Experiment.
Throughout the course of his experiment, Rutherford had his two associates (Hans Geiger and Ernest Marsden) aim a beam of alpha particles at a piece of gold foil that was approximately 8.6 x 10^(-6) centimeters thick. To be more accurate Rutherford actually included a wide variety of different foils (such as: aluminum, iron, and lead), but his use of gold foil is most commonly spoke of. In accordance to the J.J. Thomson model of an atom, the alpha particles should have passed directly through the gold foil for all instances. Therefore to confirm this activity, a zinc sulfide screen was placed behind the foil as a backdrop for the alpha particles to appear upon. Directly above this screen was a microscope that allowed one of the two experimenters to observe any contact made between the alpha particles and the screen.
After the experiment had been set up in accordance to the speculations described above, a beam of alpha particles was fired through the piece of foil and the location where the particle landed on the screen. As explained above, each particle should have gone directly through the foil if the plum pudding model was correct. For the most part, the alpha particles corresponded with this hypothesis and passed straight through the gold foil. There were, however, a small hand full of particles that deflected slightly from the straight path by about one or two degrees. But the biggest discovery was made when 1 in 20,000 particles would deflect approximately 90 degrees or more from the parent beam. In fact, an occasional particle even fired right back at the experimenter. About this, Rutherford said:
"It was as if you fired a 15-inch shell at a sheet of tissue paper and it came back to hit you."