Niels Bohr

Niels Henrik David Bohr (born 7 October 1885 - died 18 November 1962) was a Danish physicist who made important contributions to understanding the atomic structure and quantum theory for which he recieved the Nobel Prize in Physics in 1922. He was also a philosopher and promoted scientific research.
In the 1930s, Bohr helped refugees from Nazism. After the occupation of Denmark by the Nazis, he had a famous meeting with Heisenberg, who was the head of the German nuclear energy project. When word was reached that he was about to be arrested by the Germans, he fled to Sweden and then Britain where he became part of the British mission to the Manhattan Project.
In the 1930s, Bohr helped refugees from Nazism. After the occupation of Denmark by the Nazis, he had a famous meeting with Heisenberg, who was the head of the German nuclear energy project. When word was reached that he was about to be arrested by the Germans, he fled to Sweden and then Britain where he became part of the British mission to the Manhattan Project.
Why do we see colours?
In the 19th Century, scientist were sure that light was a wave with a length and a frequency. But they began to question this when light was passed through a prism and changed to 7 different colours. If light was a wave, the colours would not be there or smudged. Instead, the colours were sharp and corresponded to a particular wavelength. They began to wonder if electrons had something to do with this, but they could not imagine why.
This question interested Niels Bohr who began to research this and which led him to creating a new set of laws regarding the atom.
This question interested Niels Bohr who began to research this and which led him to creating a new set of laws regarding the atom.
Improving the Rutherford Model
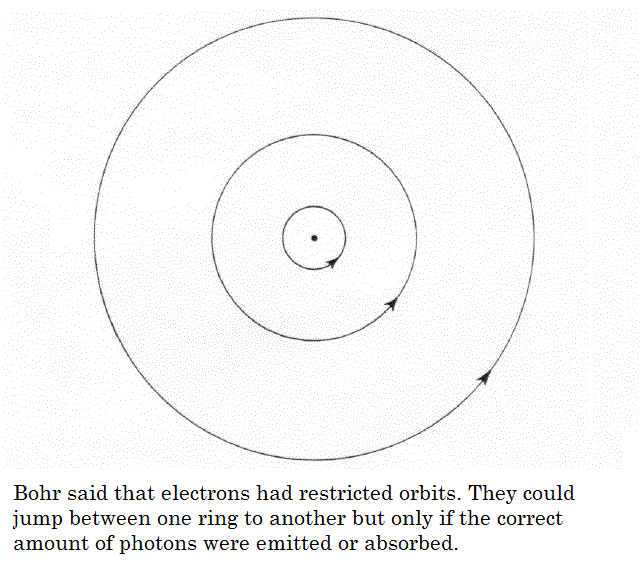
In 1913, Niels Bohr, proposed a model for the hydrogen atom for his instructor, Ernest Rutherford. and yet also explained the spectrum of the hydrogen atom. The Bohr model was based on the following assumptions.
- The electron in a hydrogen atom travels around the nucleus in a circular orbit.
- The energy of the electron in an orbit is proportional to its distance from the nucleus. The further the electron is from the nucleus, the more energy it has.
- Only a limited number of orbits with certain energies are allowed. In other words, the orbits are quantized.
- The only orbits that are allowed are those for which the angular momentum of the electron is an integral multiple of Planck's constant divided by 2.
- Light is absorbed when an electron jumps to a higher energy orbit and emitted when an electron falls into a lower energy orbit.
- The energy of the light emitted or absorbed is exactly equal to the difference between the energies of the orbits.
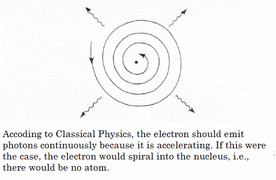
His model also explained why an atom was stable. The motion of the electrons in the Rutherford model was unstable because, according to classical mechanics and electromagnetic theory, any charged particle moving on a curved path emits electromagnetic radiation; thus, the electrons would lose energy and spiral into the nucleus. He explained radiation can occur only when the electron jumps from one orbit to another. The atom will be completely stable in the state with the smallest orbit, since there is no orbit of lower energy into which the electron can jump.
Since Niels Bohr improved Rutherford's model, this model is sometimes referred to as the "Bohr-Rutherford Model".
Since Niels Bohr improved Rutherford's model, this model is sometimes referred to as the "Bohr-Rutherford Model".
What was wrong with the Bohr Model?
|
The Heisenberg Uncertainty Principle: The Zeeman Effect |