What was the Cathode Ray Experiment?
Thomson started the Cathode Ray experiment in the 1894 at the Cavendish Laboratory at Cambridge University. He experimented with currents of electricity inside empty glass tubes. Thomson carried out three experiments to come to his conclusion.
The First Experiment
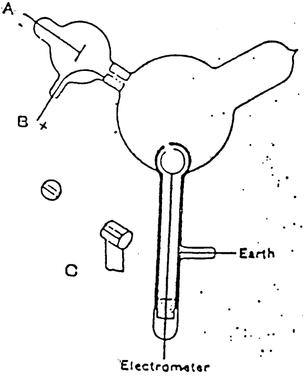
Thomson’s first experiment was to investigate whether or not the negative charge could be separated from the cathode rays by magnetism.
To observe this he built a cathode ray tube with a metal cylinder on the end. This cylinder had two slits in it, leading to electrometers, which could measure small electric charges.
Thomson found that if the rays were magnetically bent and they could not enter the slit. Therefore, the electrometer registered little charge.
Thomson concluded that the negative charge was inseparable from the rays.
He found his conclusion by applying a magnetic field across the tube. Since the electrometers recorded no activity and so the charge had been bent away by the magnet.
To observe this he built a cathode ray tube with a metal cylinder on the end. This cylinder had two slits in it, leading to electrometers, which could measure small electric charges.
Thomson found that if the rays were magnetically bent and they could not enter the slit. Therefore, the electrometer registered little charge.
Thomson concluded that the negative charge was inseparable from the rays.
He found his conclusion by applying a magnetic field across the tube. Since the electrometers recorded no activity and so the charge had been bent away by the magnet.
The Second Experiment
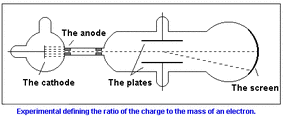
Thomson made a cathode ray with an almost perfect vacuum. This vacuum had two electrical pates halfway down the tube, one positively charged and one negatively charged. He knew that electrical charges repelled on another, which allowed him to see whether the charge was a positive or negative. He would observe this by looking at the light deflection pattern of the cathode ray
The rays were deflected by the electric charge. He concluded that cathode rays had a negative charge.
The rays were deflected by the electric charge. He concluded that cathode rays had a negative charge.
The Third Experiment
Although Thomson had performed all these experiments he was still curious to know what was the size of the cathode rays.
Thomson used a charge-to-mass ratio because he knew the weight of the cathode ray tube, the heat, the electrical current, and how much heat had been added from the electrons firing.
He concluded that the negative cathode ray particles were a thousand times tinier than an atom.
Along these lines he proved the existence of subatomic particles.
Thomson used a charge-to-mass ratio because he knew the weight of the cathode ray tube, the heat, the electrical current, and how much heat had been added from the electrons firing.
He concluded that the negative cathode ray particles were a thousand times tinier than an atom.
Along these lines he proved the existence of subatomic particles.