Ernest Rutherford
|
Ernest Rutherford, 1st Baron Rutherford of Nelson, (born 30 August 1871 - died 19 October 1937) was a New Zealand born physicist who was also considered the father of nuclear physics.
A consummate experimentalist, Rutherford was responsible for a remarkable series of discoveries in the fields of radioactivity and nuclear physics. He discovered alpha and beta rays, set forth the laws of radioactive decay, and identified alpha particles as helium nuclei. Most important, he postulated the nuclear structure of the atom. Using Radioactivity to discover the Neucleus |
In 1899 Ernest Rutherford studied the absorption of radioactivity by thin sheets of metal foil and found two components:
Before he could study the effect of -particles on matter, Rutherford had to develop a way of counting individual -particles. He found that a screen coated with zinc sulfide emitted a flash of light each time it was hit by an -particle. Rutherford and his assistant, Hans Geiger, would sit in the dark until his eyes became sensitive enough. They would then try to count the flashes of light given off by the ZnS screen.
- alpha (a) radiation, which is absorbed by a few thousandths of a centimeter of metal foil,
- beta (b) radiation, which can pass through 100 times as much foil before it was absorbed.
- Shortly thereafter a third form of radiation was discovered - gamma (g) rays that can penetrate as much as several centimeters of lead.
Before he could study the effect of -particles on matter, Rutherford had to develop a way of counting individual -particles. He found that a screen coated with zinc sulfide emitted a flash of light each time it was hit by an -particle. Rutherford and his assistant, Hans Geiger, would sit in the dark until his eyes became sensitive enough. They would then try to count the flashes of light given off by the ZnS screen.
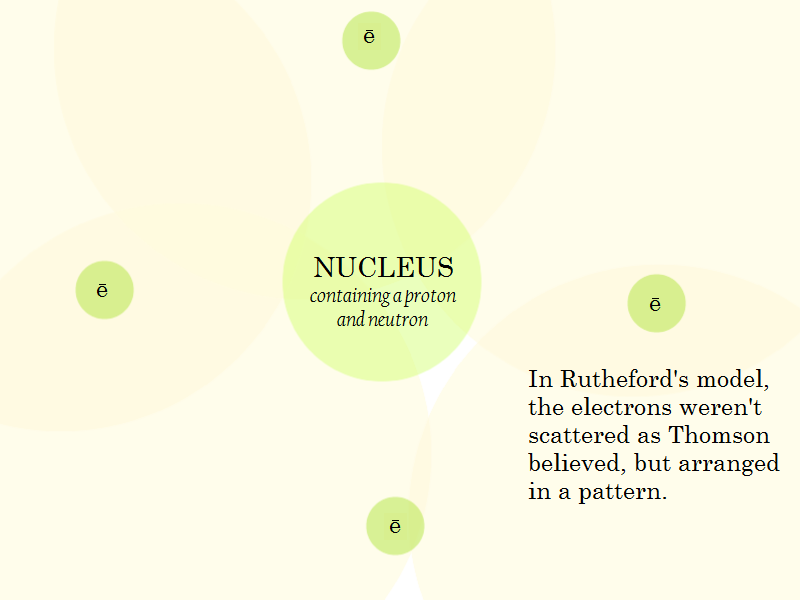
The Rutherford Model
The Proton and the Neutron
At the same time that Rutherford proposed the name proton for the positively charged particle in the nucleus of an atom, he proposed that the nucleus also contained a neutral particle, eventually named the neutron. It was not until 1932 that was able to prove that the neutral particle existed.